 Then I gave you the reason why metalloids are called semiconductors.Īnd finally we discussed the physical and chemical properties of metalloids. Then we discussed the complete list of all the metalloids with their atomic number, symbol and element name. Later on we saw that there are 6 metalloids on the Periodic table. Then we discussed the position of metalloids in the Periodic table. So in this article, we discussed the definition of metalloids. Get Info + HD Image of Periodic Table Summary You can effortlessly find every single detail about the elements from this single Interactive Periodic table. Let me tell you how this Interactive Periodic Table will help you in your studies.ġ). Free Gift for you: Interactive Periodic Table Metalloids have the property to form glasses on oxidation and so that are used in glass manufacturing.Metalloids have different metallic allotropes as well as nonmetallic allotropes.Metalloids + Halogens = Compounds (metalloids elements reacts with halogens and finally compounds are formed by this chemical reaction).Some metalloids contract when they are melted.Metalloids + Metals = Alloys (When they are mixed with metals, they forms alloys).Chemical properties of metalloidsĬhemical properties of metalloids are little bit similar to that of nonmetals. They are less conductive than metals and more conductivity than nonmetals.Physical properties metalloids are somewhat similar to that of metals. Let us discuss the physical properties as well as chemical properties of metalloids/semimetals. Hence those elements whose energy band gap lies in between the metals and insulators are known as semiconductors.Įxample: Silicon is a widely used semiconductor nowadays. Hence in semiconductors, for the electrons to jump from valence band to conduction band, they require less energy than that of insulators and more energy than that of conductors. Now in semiconductors, the band gap shown in the above image is not so high as that of insulators and not so close as that of metals.īut the band gap of semiconductors is in between the two. Atomic number Symbol Name of element 5 B Boron 14 Si Silicon 32 Ge Germanium 33 As Arsenic 51 Sb Antimony 52 Te Tellurium Why are metalloids called semiconductors? Here is a complete list of metalloids on the Periodic table. Let us now see the list of metalloids/semimetals with their atomic number, symbol and name. Hence, there are total 6 known metalloids/semimetals on the Periodic table. And in chemistry, the chemists are concerned with the chemical properties of metalloids.)Īlso, Polonium and Astatine are synthetic elements and they have very short half life. In physics, they may define metalloids on the basis of physical properties. (That means in metallurgy, the researchers may define metalloids on the basis of density. (Source: HRW texts)Īlso this number is inexact due to lack of universally accepted definitions. 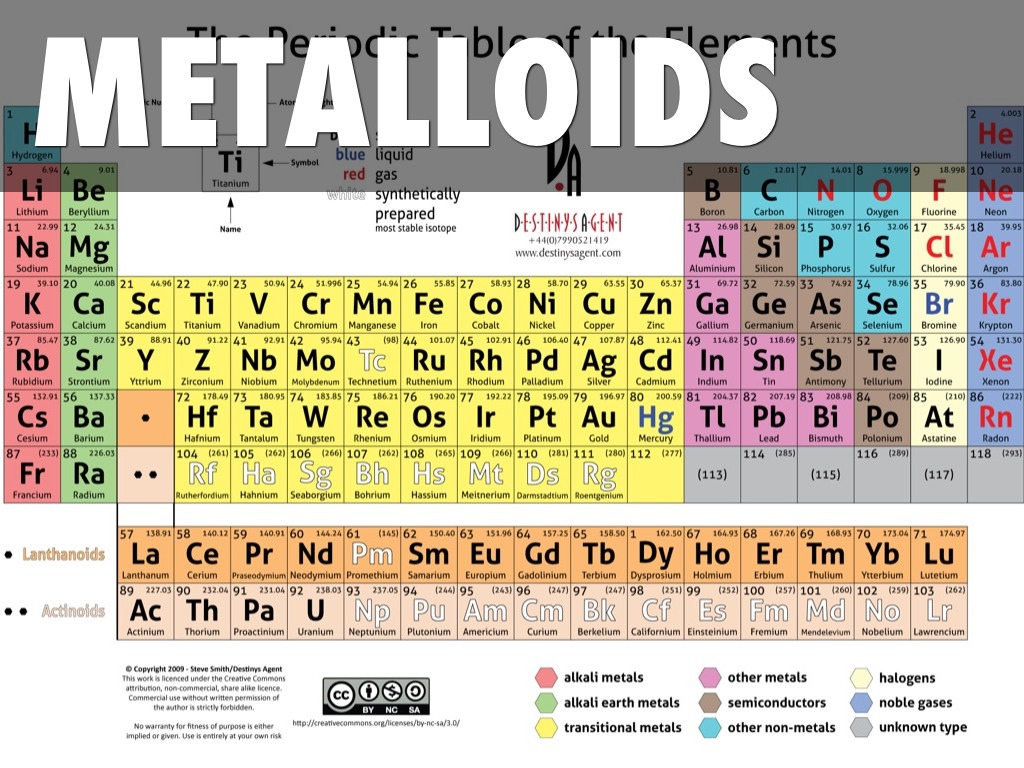
Thus Polonium and antimony are not included in the category of metalloids by many researchers. And Astatine shows more characteristics of nonmetals (halogens). Many researchers have found that Polonium shows more characteristics of metals. Should we consider Polonium and Astatine as metalloids? These two elements which show fewer characteristics of metalloids are: These elements which shows maximum characteristics of metalloids are:īut the other two elements also show few characteristics of metalloids and they are represented in green color on the above Periodic table. Why are metalloids called semiconductors?Īs you can see in the above image, there are six known metalloids which show maximum characteristics of the metalloids and they are represented in orange color.List of metalloids on the periodic table.How many metalloids are there in the Periodic table?.If you want to skip to any part of this article, just click on the below links !!! I have solved all those doubts in this single small article. I hope you have got the answer of “Where are Metalloids located on the periodic table?”īecause there are lot more things you need to know about the metalloids and most of the students have confusion regarding the total number of metalloids present on the Periodic table. In other words, metalloids (semimetals) are located on the right side of the post transition metals and on the left side of nonmetals (see above image).Īlso we can say that metalloids are present in the diagonal region of the p block on Periodic table. 
They form a separating boundary between the metals and nonmetals. The orange color on the Periodic table represents metalloids. Metalloids are located between the metals and nonmetals.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
